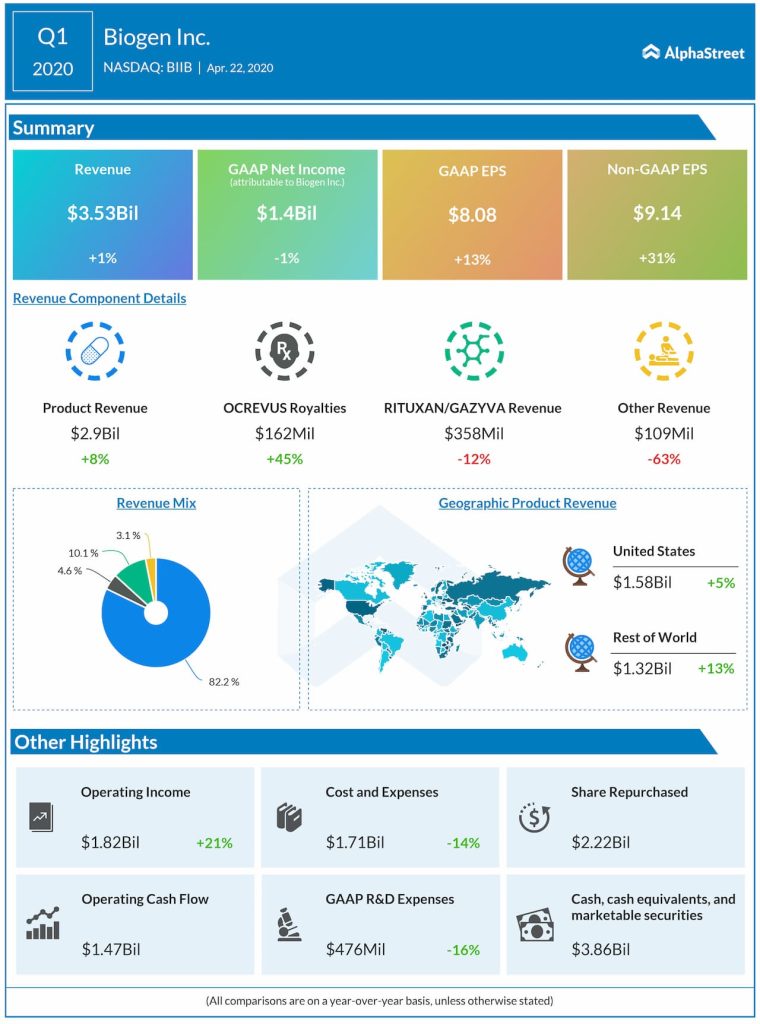
Late last year, Biogen (NASDAQ: BIIB) made an announcement that left investors gung-ho. The stock soared 27% after the company optimistically said it would file an application with the US Food and Drug Administration for the approval of its Alzheimer’s candidate, aducanumab.
However, during the first-quarter earnings conference call, the company revealed that the filing would be delayed by at least a couple of months, and was now expected around the third quarter. Lack of clarity in the reasons presented by the management left analysts visibly befuddled during the entirety of the call duration.
Only one topic
All the 11 questions asked by analysts during the Q&A session revolved around the subject of aducanumab filing as analysts kept digging to find clarity. It may be noted that the drugs that are currently available in the market for Alzheimer’s help only in alleviating the symptoms, unlike aducanumab, which treats the disease. Over 5.5 million Americans are estimated to be suffering from this progressive brain disorder, which erases memory and destroys mental functions.
Without going into specifics the company maintained that the original timeline of early 2020 was a rough estimate based on the progress they were seeing at that point in time. While COVID-19 was a contributing factor to the delay, it wasn’t the only one. Biogen’s Executive Vice President of Research & Development Alfred Sandrock also blamed the complicated process:
“Timing is not easy to predict. We were trying to do that since the initial announcement in October. And this is an unusual process. But I want to emphasize that we now have an open BLA. We’ve started to submit modules. We have continued constructive engagement with FDA and we believe we’re on track in terms of the potential for approval.”
The executive added that there were no changes in the interpretation of the data, insisting that the additional meetings were in the pipeline to engage better with the FDA.
[irp posts=”57665″]
Employees affected
Biogen management said its employees were affected by COVID-19, but all of them have either recovered or are in the process of recovering. Sandrock said, explaining the role of the pandemic in the delay when it seemed analysts were not leaving the topic behind:
“Some members of the team did get COVID and I can tell you, it’s hard to work when you have COVID. The fatigue — mental and physical fatigue was such that there were some people who were affected by the disease.”
Both analysts and investors were clearly underwhelmed by the response. BIIB stock has declined 11% since the earnings announcement, even though it surpassed analyst projections.
The company added that samples and medical data were collected from volunteering employees who recovered from COVID-19, as well as their contact, which will be included in the recently launched biobank.
[irp posts=”56002″]
Biogen had earlier this month announced
the launch of the COVID-19 biobank along with the Broad Institute of MIT and
Harvard and Partners Healthcare. Speaking about the biobank, Sandrock said:
“This biobank aims to centralize and facilitate study of a large collection of the identified samples and medical data with the aim of unraveling the biology of COVID-19, linking molecular signatures with clinical presentation and accelerating the search for potential treatments.”
For more updates about the company, read the full transcript here.