Krystal Biotech, Inc. (NASDAQ: KRYS), a leader in redosable gene therapies, today announced record-breaking financial results for the fourth quarter and full year ending December 31, 2025. Driven by the aggressive commercial uptake of VYJUVEK® and significant pipeline milestones, the company reported a substantial increase in both top-line revenue and net income.
Financial Performance Milestones
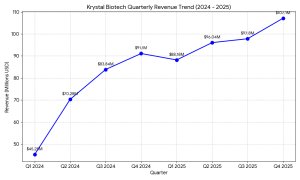
- Record Quarterly Revenue: Net product revenue for Q4 2025 reached $107.1 million, an 18% increase over the same period in 2024. This exceeded the consensus analyst estimate of $106.16 million, marking a strong finish to the fiscal year.
- Annual Growth Trajectory: Full-year 2025 revenue surged to $389.1 million, representing a 34% year-over-year increase from the $290.5 million reported in 2024.
- Surging Profitability: Quarterly net income was $51.4 million, or $1.70 per diluted share, beating the forecast of $1.58. For the full year, net income reached $204.8 million ($6.84 per diluted share), more than doubling the $89.2 million reported in the previous year.
- Industry-Leading Margins: The company maintained a consistent 94% gross margin on VYJUVEK throughout 2025, underscoring the efficiency of its proprietary manufacturing platform.
- Robust Liquidity: Krystal closed the year with $955.9 million in cash, cash equivalents, and investments, providing a formidable capital base for its expanding clinical pipeline.
Global Commercial & Pipeline Momentum
- VYJUVEK Market Penetration: In the United States, Krystal has secured over 660 reimbursement approvals. The therapy’s footprint is expanding internationally, with launches now active in Germany, France, and Japan, where approximately 90 patients have already been prescribed the treatment.
- Oncology Breakthrough: In February 2026, the FDA granted Regenerative Medicine Advanced Therapy (RMAT) designation to KB707 for advanced non-small cell lung cancer (NSCLC), an accelerator aimed at shortening the path to potential approval.
- Respiratory Progress: The company recently confirmed successful lung delivery of the CFTR protein in its Phase 1 study of KB407 for cystic fibrosis. Enrollment for the registrational CORAL-3 study is slated to begin in the first half of 2026.
- Dermatology Expansion: KB111 (Hailey-Hailey disease) received FDA Fast Track Designation in January 2026, while KB105 is expected to enter Phase 2 pediatric trials later this year.
2026 Strategic Outlook
- Expense Guidance: Management anticipates 2026 non-GAAP operating expenses (R&D and SG&A) to fall between $175 million and $195 million. This reflects increased investment in global commercial infrastructure and the advancement of multiple registrational trials.
- Expansion Targets: The company aims to launch VYJUVEK in at least one additional major European market and expand its specialty distributor network to cover over 40 countries by year-end.
- Near-Term Catalysts: Multiple registrational data readouts are anticipated before the end of 2026, specifically for the KB803 (ocular DEB) and KB801 (neurotrophic keratitis) programs.
“2025 was a transformative year that validated the global demand for redosable gene therapy,” said Krish S. Krishnan, Chairman and CEO. “With a strong cash position and a platform demonstrating versatility across skin, lung, and eye tissues, we are entering 2026 with unparalleled momentum.”
