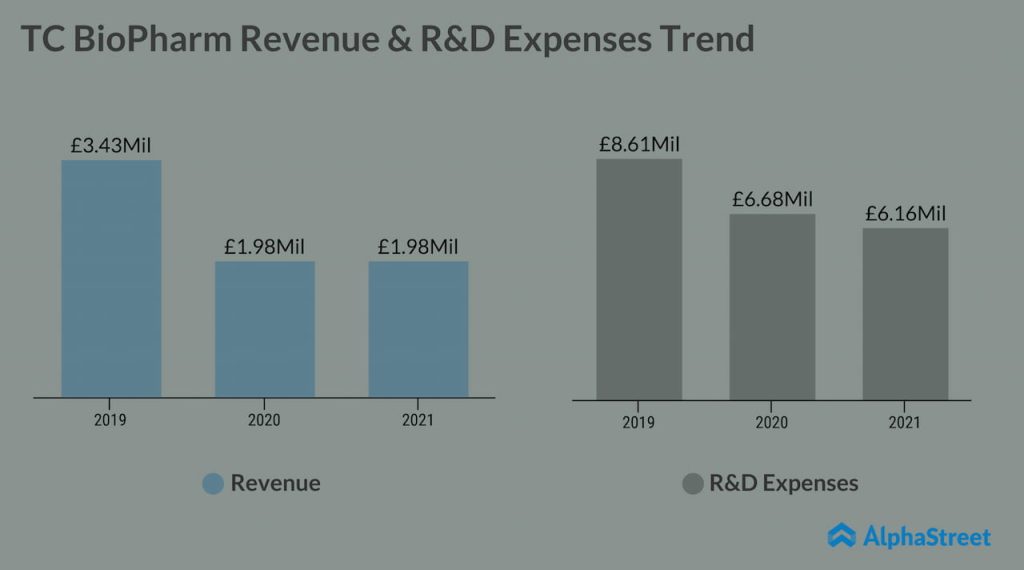
TC BioPharm (NASDAQ: TCBP) is a clinical-stage cell therapy company focused on the development of treatments for infectious diseases, including advanced allogeneic chimeric antigen receptor (CAR) T-cell therapy products for the treatment of cancer. Established in 2014 in Edinburg, Scotland, the company has recently reorganized and re-focused its efforts on gaining U.S. FDA approval for OmnImmune, an unmodified allogeneic gamma delta T-cell product initially targeted for the treatment of Acute Myeloid Leukemia.
In an email conversation with AlphaStreet, TC BioPharm’s chief executive officer Bryan Kobel provided insights into the company’s clinical programs and future plans:
Can you explain the importance/differentiator of your platform allogenic Gamma Delta T-Cell Based Immunotherapy?
We are developing safer, less expensive products to target more cancers and save more lives – particularly allogeneic therapies as the future of cancer and infectious disease treatment. The key significance of allogeneic products is their ability to be stored and delivered point of care, or in short need, versus a product that is derived from the patient. Additionally, and this is core to oncology patients, cancer suppresses the immune system and weakens your system greatly. So an allogeneic approach allows us to take healthy cells, activated cells ready for a fight, from healthy donors and give them to sick/unhealthy patients in a timely fashion.
OmnImmune is based on our platform technology for allogenic therapies that enable us to create and bank donor T-cells, which differs from an autologous approach that uses a patient’s own blood for T-cells. The platform allogeneic gamma-delta T-cell therapies we’re developing for cancer indications have enabled us to show lower-cost and scalable human efficacy data in Acute Myeloid Leukemia (AML) with reduced dependence on the fitness of a patient. At the core, gamma-delta T-cells become less potent and less prevalent as people age, become sick, or immuno-compromised. Our ability to manufacture young, active gamma-delta T-cells using donor blood enables us to provide billions of healthy gamma-delta T-cell populations for cancer patient infusions.
Clinical development to date has enabled us to take a logical, safer, and more agile approach to our clinical trials. By starting with an autologous, unmodified gamma delta product, we were able to establish safety prior to entering the clinic in 2018 with our unmodified allogeneic product. Now, our focus is on clinical development and refinement of products based on the ideal GDT vehicle with a proprietary genetic CAR-T arsenal to fight a range of cancers.
How is the clinical program on TCB-008 progressing?
The past three months have been incredibly exciting and we’re moving quickly down several tracks toward the commercialization of cell therapies. Foremost have been the steps we’ve taken to re-focus our clinical strategy on U.S. FDA trials for OmnImmune, also known as TCB-008.
Over the last 12 months, we have created multiple research collaborations and strategic relationships, the majority of which are U.S.-based and beginning to come to fruition. Another key element in this strategy was adding Bree Harlin to our management team as Chief Clinical Officer to lead our Global Clinical Division with a focus on managing U.S. clinical trials. She joined the team from Loxo at Lilly, where she worked on the clinical development of targeted therapies for various types of solid and hematologic cancers. She has worked in clinical development and operations across multiple therapeutic areas at large pharma and small biotech companies for most of her career which began in academia working on clinical trials at Mass General and Brigham and Women’s Hospital.
With Bree on the team, in February we announced the dosing of our final patient from our phase 2B clinical trial of OmnImmune. TC BioPharm’s trial involved administering TCB-008 to diagnosed patients with safety review by an oversight board. Completing the dosing of the safety cohort was another step in our efforts and firmly plant TCBP as the leader in the allogeneic gamma delta space.
With regard to your initiatives on future FDA trials for TCB-008, what are the different stages and the tentative timeframe for approval?
The pending protocol submission will be a Phase 1b safety trial, with a relatively small patient population and a short timeline to completion. The first step is filing an Investigational New Drug (IND) Application with the FDA, which we anticipate for the third quarter of 2023. This trial will most likely be a dose-escalating trial consisting of 9-12 patients, a safety trial that we hope would show indications of efficacy. The trial is a departure from our ACHIEVE trial in the sense we are ramping up the dosing substantially, the lowest dose is expected to be around 700m cells, and the highest around 2.2 billion. We expect an additional IND to be filed in early 2024 as well.
Prioritizing US trials realigns us with our long-term goal of becoming a leading commercial-stage company, with a myriad of oncological treatment applications for TCB-008 as both a monotherapy and as a combination therapeutic. We firmly believe in the potential of our asset and the best way to position the company for success is to commence with this proposed US trial protocol and to pursue future trials through the FDA pathways. TC BioPharm expects this US clinical trial enrollment to be relatively rapid due to the fact that America offers a significantly greater pool of patients with more than 20,000 AML diagnoses each year. This transition will allow TC BioPharm to become more economically efficient by simplifying its strategy and reducing manufacturing and production efforts.
Can you provide details of the ongoing partner programs?
We’re now better positioned for near-term success and sustainability, including the potential for key data readouts in 2024 stemming from the partnership we announced in January with The University of Texas MD Anderson Cancer Center. That is a highly valuable component for our FDA trials. We’re excited about our new collaboration as an opportunity to expand the knowledge base of how gamma-delta T-cells work in oncology settings. This collaboration brings together MD Anderson’s clinical trial infrastructure and the translational research expertise of its immunotherapy platform with the clinical data of TC BioPharm’s targeted gamma-delta T-cell oncology pipeline.
Our goal is to have our research teams work together on both preclinical and clinical studies to evaluate the potential for gamma-delta T-cell therapies and to better understand the behaviors of these cells in patients. Ultimately, a steering committee will drive the development of both the data set and subsequent efficacy assessments for each study. This is an incredible combination of comprehensive capabilities to uncover detailed insights into the behavior of gamma-delta T-cells and corresponding responses in patients. We expect this to be a large part of what happens next with TCB-008 as well as to help us to better design targeted cancer studies in additional blood cancers.
Do you see the need for external funding as the clinical programs enter advanced stages?
External funding and cost savings are critical to getting us closer to the finish line. External funding will help us steer through current market realities and macro headwinds facing biotechnology companies, as will our plans to protect TC BioPharm’s long-term sustainability and ensure our growth plans are properly capitalized. Earlier this month we announced a restructuring for cost savings in excess of $3 million on an annual basis. This is tethered to our focus on U.S. clinical trials to help reduce cost structure and streamline operations. As we continue to raise external funding, TCBP will continue to consider additional cost savings programs and non-dilutive funding options as well as strategic collaborations and partnerships. Cancer is an expensive disease to treat, so it’s an expensive drug development process, but we see TCB-008 as potentially lowering the cost of treatment substantially at scale.
External funding, realigning our resources, and restructuring the organization will help us focus on the critical programs that are expected to improve the lives of patients and establish long-term value for shareholders.