Eli Lilly & Co.’s (NYSE: LLY) shares have
gained 29% since the beginning of the year and 16% over the past three months. The
sentiment around the stock remains bullish after the company announced the world’s
first study of a potential antibody treatment to fight COVID-19.
Through its collaboration with AbCellera, Lilly developed the
investigational drug named LY-CoV555, which is specifically designed to attack
the novel coronavirus. On Monday, the company announced that the first patients
in the study were dosed.
Lilly has been looking at multiple ways to treat COVID-19, including studying its existing drugs and partnering with two biotech companies to discover novel antibody treatments. The company is looking at both single antibody therapy as well as antibody cocktails for the treatment of COVID-19.
There is significant opportunity in the COVID-19 treatment space
and any positive development in this area could provide a huge boost to the
company’s growth and stock. Rivals like Gilead (NASDAQ: GILD) and Moderna
(NASDAQ: MRNA) have also
recently published data and moved on to the next phase of their coronavirus
vaccine studies.
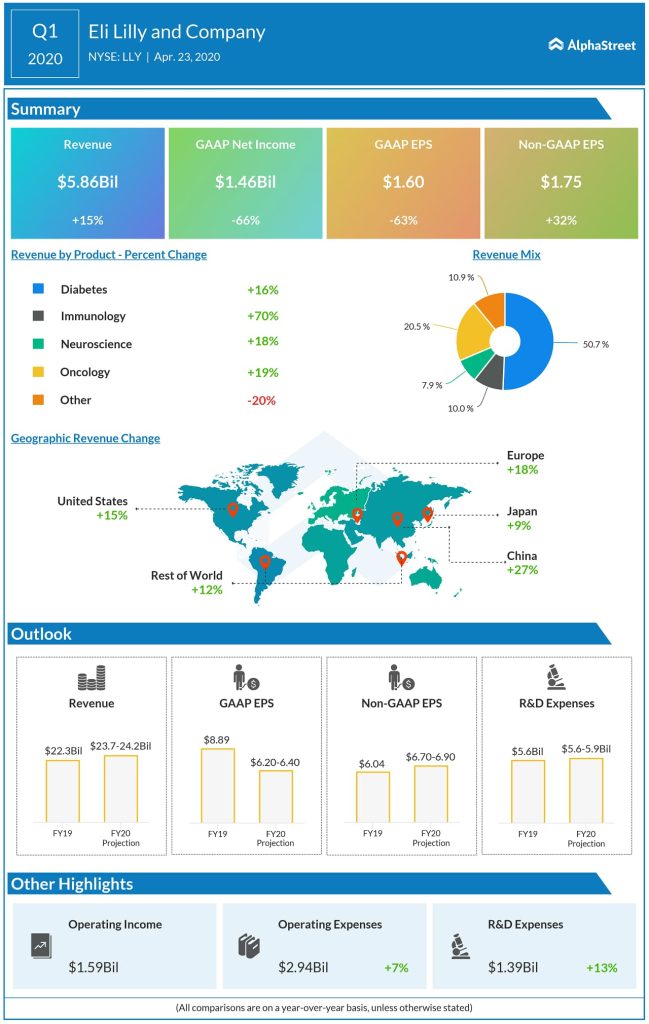
In its most recent quarter, Lilly saw 15% revenue growth
along with 22% volume growth. The company generated around $250 million in
additional revenue due to increased customer buying patterns and patient
prescription trends due to the pandemic.
“Lilly exited 2019 with strong revenue growth and margin expansion, driven by the uptake of our newer medicines. That momentum continued in Q1 2020 and was augmented by higher patient and supply chain purchasing due to the COVID-19 pandemic.” – Josh Smiley, CFO
Lilly’s strong product portfolio of successful drugs that scale
across several medical conditions provide the company with a strong advantage. Key
growth products including Trulicity, Taltz and Jardiance contributed 19% of
revenue growth in the first quarter of 2020.
In terms of its COVID-19 treatment efforts, the company is
studying the potential use of baricitinib, which is its JAK inhibitor in
collaboration with Incyte, to treat the virus and expects to see the results in
a month or two.
Lilly is also undertaking a Phase 2 trial with a monoclonal
antibody against Angiopoietin 2 in pneumonia patients with COVID-19 who are at
risk of progressing to acute respiratory distress syndrome and expects the
results over the coming months.
Despite the near-term benefits and long-term opportunities from
COVID-19, the company faces challenges as well from the pandemic. Further into
the year, Lilly expects headwinds from destocking due to a normalization in
supply chain after the recent demand surge, a drop in new prescriptions and
pricing pressures.
Keeping these challenges in mind, Lilly has guided for reported EPS of $6.20-6.40 and adjusted EPS of $6.70-6.90 for the full year of 2020. Revenues are expected to be $23.7-24.2 billion.
Click here to read the full transcript of Eli Lilly Q1 2020 earnings call