Medtronic plc (NYSE: MDT) is scheduled to report second quarter 2020 earnings results on Tuesday, November 19, before the market opens. The consensus estimate is for earnings of $1.28 per share on revenue of $7.66 billion.
The momentum seen in the Minimally Invasive Therapies Group and Restorative Therapies Group is expected to continue and in turn fuel the topline numbers. The weakness in the Cardiac and Vascular Group is expected to continue. The company has been making progress with several product launches and partnerships during the quarter.
In October, the company received Breakthrough Device designation from the FDA for its Valiant TAAA Stent Graft System which is intended for the repair of thoracoabdominal aortic aneurysm (TAAA) through a minimally invasive procedure. TAAA causes the bulging of the aorta, which extends from the chest into the abdomen.
Medtronic also launched the Valiant Navion thoracic stent graft system in Japan for the minimally invasive repair of thoracic descending aortic aneurysms (TAA) and complicated type B aortic dissections (TBAD).
Also see: Medtronic Q1 2020 Earnings Conference Call Transcript
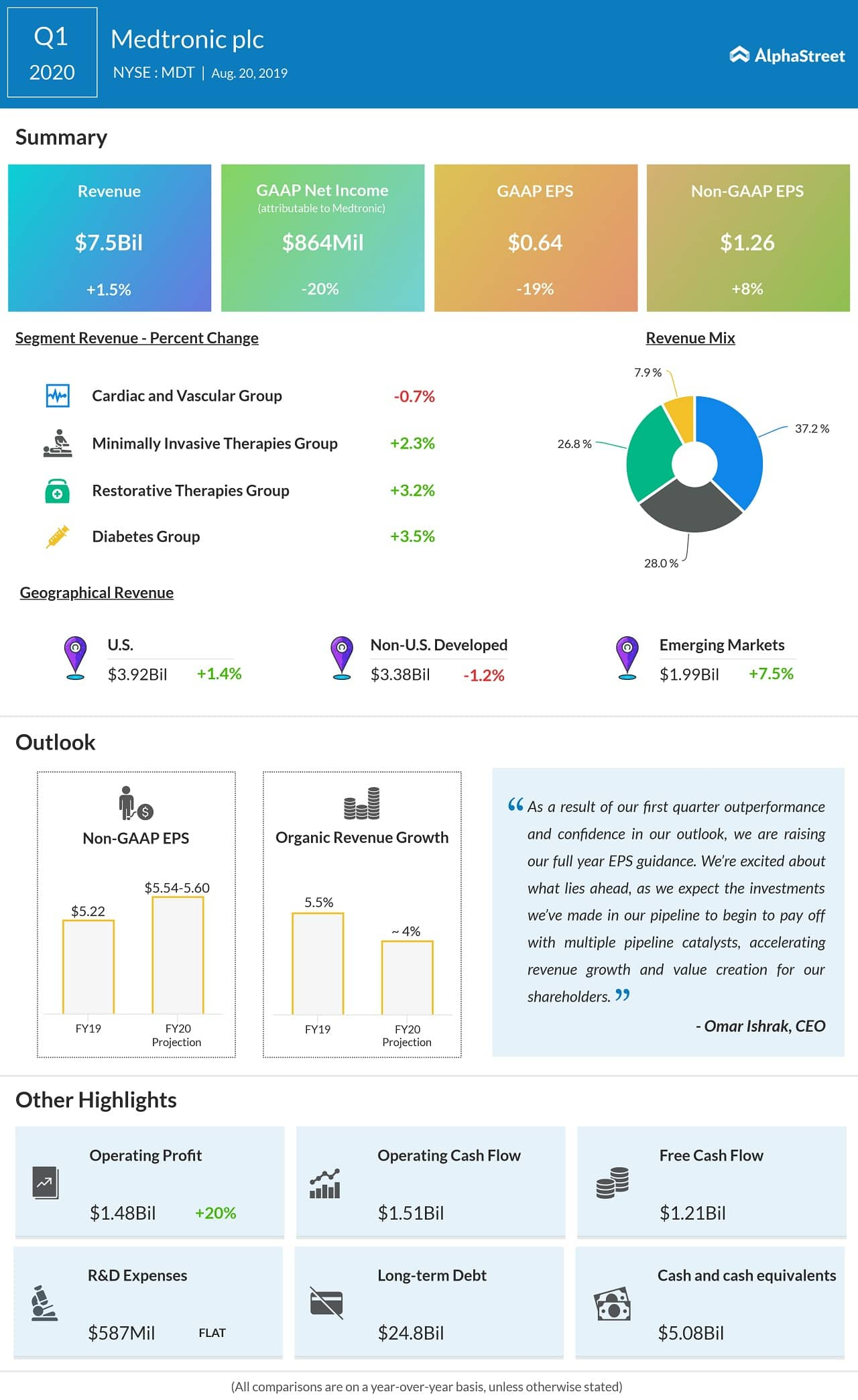
In the first quarter of 2020, Medtronic beat revenue and earnings expectations. Revenue grew 1.5% to $7.49 billion while adjusted EPS rose 8% to $1.26 compared to the year-ago quarter.
For fiscal year 2020, Medtronic expects revenue to grow 4% and adjusted EPS to come in the range of $5.54 to $5.60.
Shares of Medtronic have gained 21% so far this year and 2% over the past one month.