Alexion Pharmaceuticals, Inc. (NASDAQ: ALXN) reported its financial results for the quarter ended March 31, 2020, on Wednesday before the market opens. The results exceeded analysts’ expectations.
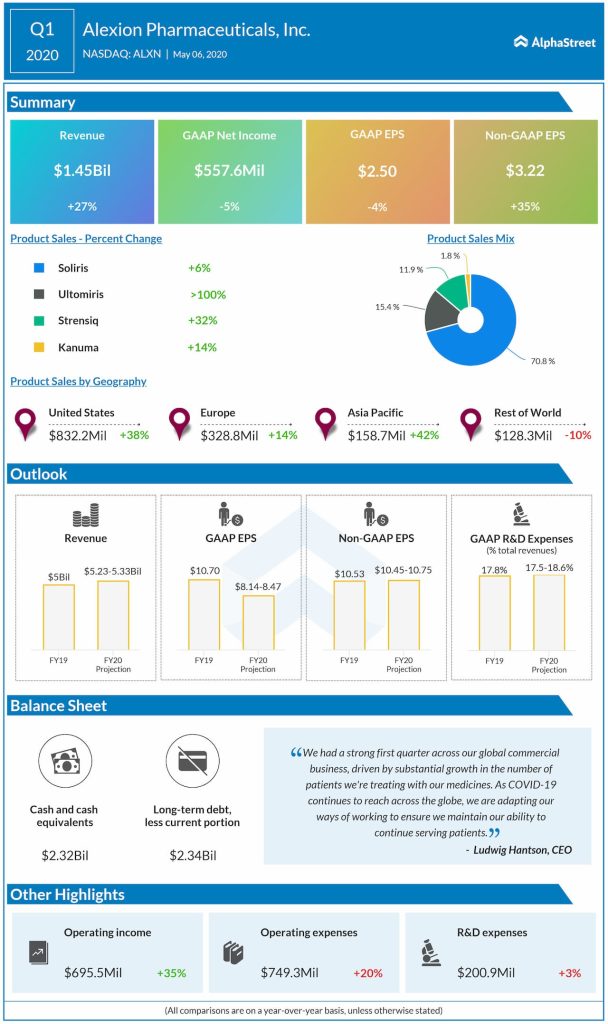
Alexion posted a 5% decline in earnings for the first quarter of 2020 from last year, which included deferred tax benefits and tax elections related to intellectual property and release of an existing valuation allowance. The top-line jumped by 27% backed by the substantial growth in the number of patients.

While the COVID-19 impact varies by study and program, generally, the company expects there will be little timing impact on fully-enrolled trials and a timing shift of at least three months on trials. The company is exploring the potential role of Ultomiris and Soliris for the treatment of severe COVID-19 and has recently initiated a Phase 3 randomized controlled trial of Ultomiris in a subset of adults with coronavirus.
Looking ahead into the full-year 2020, the company lowered its total revenues outlook to the range of $5.23-5.33 billion from the previous range of $5.5-5.56 billion, and its adjusted EPS guidance to the range of $10.45-10.75 from the prior range of $10.65-10.85. The GAAP EPS forecast is tightened to the range of $8.14-8.47 from the prior range of $7.91-8.71.