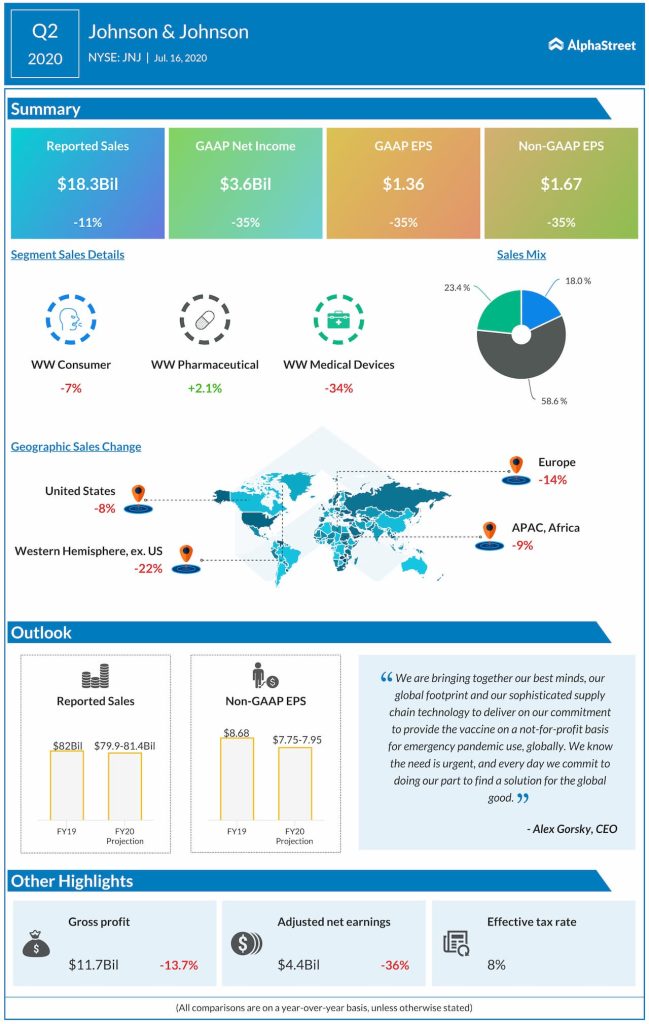
Johnson & Johnson (NYSE: JNJ) saw revenue and earnings decline for the second quarter of 2020 but both the top and bottom line numbers came in better than analysts’ projections. The company raised its guidance for the full year of 2020 based on strength in the pharmaceutical division, which was the only unit to register an increase in sales during the quarter. After staying in red territory for most of the day on Thursday, the stock saw an uptick during afternoon hours.
Quarterly performance
During the second quarter, J&J’s medical devices unit took the biggest hit, dropping nearly 34% on a reported basis. The main reason for the decline was the postponement of elective surgeries during the quarter due to the coronavirus outbreak. In orthopaedics, declines were visible in hips, knees and spine. Lower levels of travel, recreation and general activity led to a reduction in trauma.
Surgery saw declines in both the general and advanced categories, with endocutters and biosurgery seeing double-digit declines. In vision, contact lenses saw less consumption by existing users and lower number of new users due to the ongoing health crisis.
In the consumer health segment, declines in beauty, baby care, women’s health and wound care were offset by growth in OTC and oral care. The OTC segment benefited from an increase in market and share growth in Pain due to the pandemic while oral care gained from increased demand for LISTERINE mouthwash.
The pharmaceutical segment saw total sales growth of 2.1% during the quarter on a reported basis, with a nearly 6% gain in the US offsetting a 2.4% decline in international. The company saw growth in immunology, which was driven by strength in STELARA in Crohn’s Disease and ulcerative colitis as well as TREMFYA in psoriasis. Infectious diseases and neuroscience witnessed growth fueled by strength in SYMTUZA and INVEGA while oncology saw gains in DARZALEX and IMBRUVICA.
COVID-19 vaccine
Last month, J&J announced that it accelerated the initiation of its Phase 1/2a first-in-human clinical trial for its COVID-19 vaccine, which was earlier set for September, to the second half of July. On Thursday, during its Q2 earnings conference call, the company said it plans to start a late-stage human trial for the vaccine ahead of schedule in late September.
“Thanks to the tireless work of our colleagues around the world and our broad range of capabilities, we continue to successfully navigate the external landscape, and we remain focused on advancing the development of a vaccine to help address this pandemic and save lives.” – Alex Gorsky, Chairman and CEO
The Phase 1 human trial scheduled to start next week will include more than 1,000 participants. J&J is also planning to conduct a Phase 2 study in the Netherlands, Spain and Germany. The company is also working on expanding its manufacturing capacity worldwide, and if the vaccine proves to be safe and effective, J&J is looking to supply over 1 billion doses globally through the course of 2021.
Pipeline
On Tuesday, the US FDA approved TREMFYA for the treatment of active psoriatic arthritis in adults. J&J also received approval from the European Commission for its Ebola vaccine regimen earlier this month. The company’s subsidiary Janssen Pharmaceutical is collaborating with WHO on facilitating broader access to the vaccine.
Click here to get access to the transcript for Johnson & Johnson Q2 2020 earnings conference call