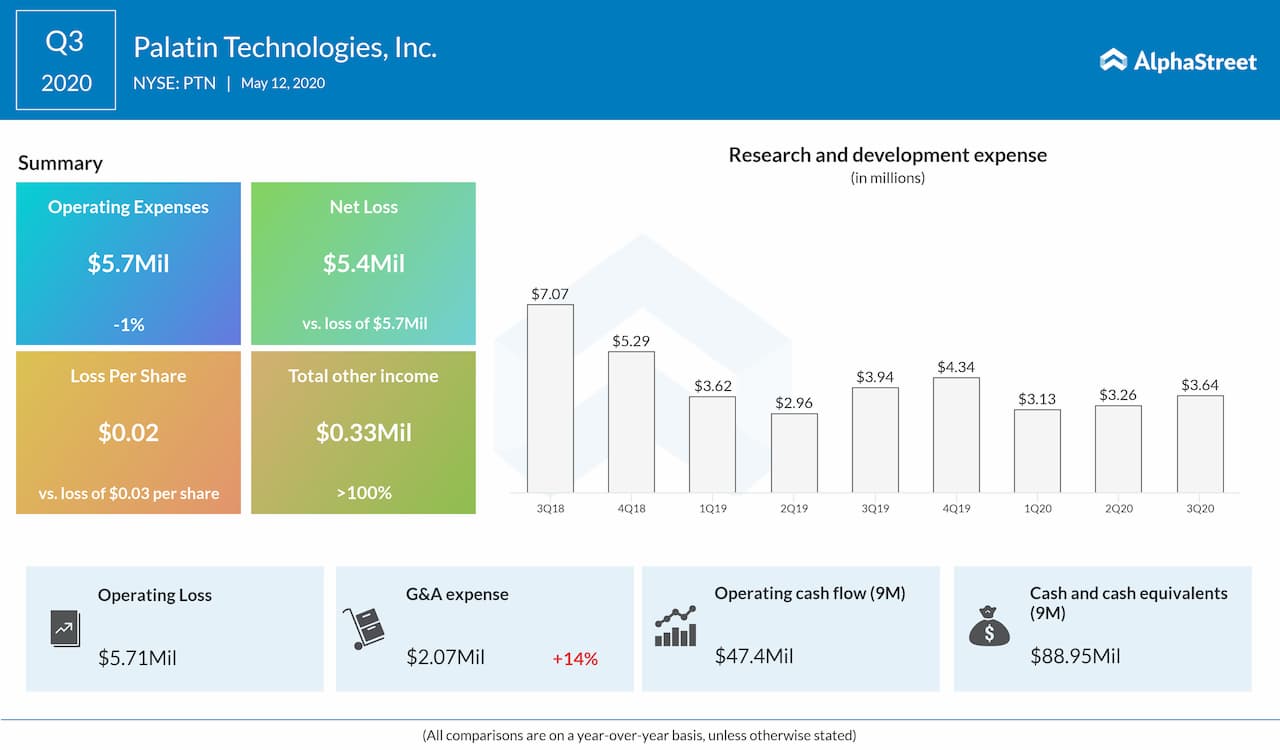
Palatin Technologies, Inc. (NYSE: PTN) reported a narrower loss in the third quarter of fiscal 2020 helped by lower operating expenses as well as higher other income. The bottom line came in line with the analysts’ expectations.
The company has implemented multiple measures in response to the COVID-19 pandemic to safeguard the health and well-being of employees while continuing to advance its programs. The company anticipates restarting enrollment of phase 2 clinical study of dry eye disease in June 2020 with the data readout is targeted for Q4 of calendar 2020.
Palatin now expects to start a phase 2 proof-of-concept clinical study with an oral formulation of PL8177 in ulcerative colitis patients in the first half of the calendar 2021, with data readout in the first half of the calendar 2022. The company continues its assessment and development work related to the treatment of patients with diabetic retinopathy, with an IND targeted for mid-calendar 2021.
The company now anticipates filing an IND and commencing clinical trials with PL8177 for non-infectious uveitis, for which FDA granted orphan drug designation, in the second half of the calendar 2021. Palatin continues to closely monitor AMAG’s process related to the divestiture of Vyleesi and AMAG’s obligations under the Vyleesi license agreement.
Take a look at our Health Care articles here
